NATURAL CYCLES
Nutrient Cycling - the Biogeochemical Cycles
|
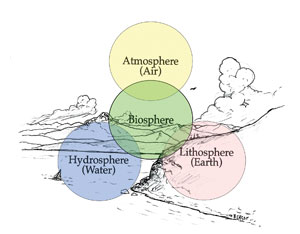
| Figure 2.24: Four Spheres. Source: Todd Berget |
Nutrient cycles are systems by which substances (chemical elements or molecules) move through the biotic (living) compartment of the earth—or the biosphere— as well as the three abiotic (non-living) components of the earth: the lithosphere (crust and upper mantle of the earth), the atmosphere (the gasses surrounding the earth), and hydrosphere (the water of the earth). Through this biogeochemical cycling, nutrients such as oxygen, carbon, nitrogen, phosphorous, and sulfur are used and recycled. Each of these cycles is complex. These brief descriptions and diagrams will provide a general overview of how these systems work and why they are important.
Four Spheres
Scientists classify the living and non-living material of the earth into four spheres (Figure 2.24).
The lithosphere includes the entire crust of the earth. “Litho” is the Greek word for stone. From the highest peak in Waterton National Park to the rich soil of the Mission Valley, this is all part of the lithosphere.
The hydrosphere includes all of the water of the earth. “Hydro” is the Greek word for water. The glaciers of Glacier National Park, lakes, rivers, and streams—and even the moisture in the air is part of the hydrosphere.
The atmosphere is the air around us. “Atmo” is the Greek word for air. The atmosphere is made up of 79% nitrogen and almost 21% oxygen: the rest is carbon dioxide and other gasses.
The biosphere is composed of the living organisms. “Bio” is the Greek word for life. The biosphere includes all life, from the single-celled organisms in a pond to the mighty grizzly bear, and from tiny lichens to Douglas-fir trees.
These four spheres are interconnected. As an example, a bull trout (part of the biosphere) swims upstream (part of the hydrosphere) to spawn. Some water from the stream (hydrosphere) trickles through the rock and soil (part of the lithosphere).
Changes to spheres can be natural (earthquake) or human caused (air pollution), and changes in one sphere often result in changes in another. Changes can be local (a flood may spread only a few miles) or widespread (the change in ocean currents from an El Nińo event can change the weather across a continent). The four spheres work together to create our environment and sustain life on earth. Within the environment are the smaller systems called ecosystems.
Oxygen Cycle
Most of the earth’s oxygen is contained within the rock materials of the earth’s crust and mantle. A very small amount of free oxygen exists in either the biosphere or atmosphere. The greatest source of atmospheric oxygen is plant photosynthesis which produces sugars and oxygen from carbon dioxide and water. Photosynthesizing organisms include plants on land and in the water. Photosynthesis maintains the level of oxygen in the atmosphere and provides energy to all life forms—either directly (for the plants themselves) or indirectly (as the food source for organisms that consume the plants, and, ultimately the organisms that consume them). Additional atmospheric oxygen is the result of photolysis, a breaking down by ultraviolet radiation of atmospheric water and nitrite into component atoms. The atmosphere loses oxygen as organisms respire and decay, methods by which bacteria and animal life consume oxygen and release carbon dioxide.
Carbon Cycle
One of the most important cycles on earth, the carbon cycle is the process through which the organisms of the biosphere recycle and reuse carbon. Carbon moves through four interconnected reservoirs: the atmosphere, the terrestrial biosphere (including non-living organic materials and freshwater systems), the oceans, and sediments (including fossil fuels). Carbon moves between these reservoirs as a result of biological, chemical, physical, and geological processes. The ocean holds the largest active reservoir of carbon near the earth’s surface. Forests store 86% of the above-ground carbon and 73% of the soil carbon. Carbon is released into the atmosphere through plant and animal respiration; the decay of plant and animal material; the burning of fossil fuels such as coal, oil, and natural gasses; volcanic eruptions; and in reactions at the surface of the oceans.
 |
| Figure 2.25: Carbon cycle. Source: Author- Cunningham, W., et. al. 2007; Publisher - McGraw-Hill, N.Y. with permission of The McGraw-Hill Companies. Numbers indicate approximate exchange of carbon in gigatons (Gt) per year. Natural exchanges are balanced but antthropogenic (human caused) sources product a net increase of CO2 in the atmosphere. |
Nitrogen Cycle
Nitrogen, the most abundant element in the atmosphere, is essential to all life. It is necessary for numerous biological processes, and is a critical component of Deoxyribonucleic Acid (DNA), the genetic instructions for all living things and Ribonucleic acid (RNA), biologically important molecules. Nitrogen is plentiful in the earth’s atmosphere, but not in a form plants can use. Nitrogen must be converted from its gaseous form (“fixed” into nitrogen compounds such as nitrate) to be utilized by living organisms. The amount of fixed nitrogen determines how much food can be grown in a terrestrial environment, so it is—along with a combination of other nutrients—a critical consideration in food production.
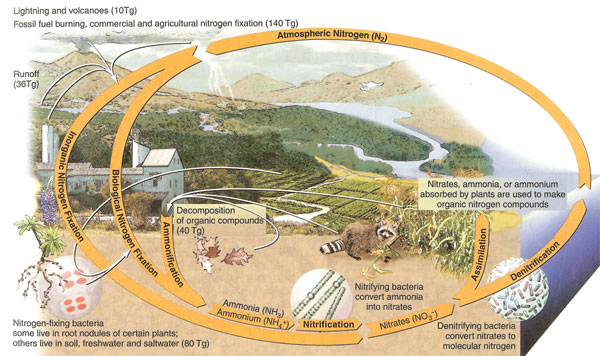 |
| Figure 2.26: Nitrogen cycle. Source: Author - Cunningham, W., et. al. 2007; Publisher - McGraw-Hill, N.Y. with permission of The McGraw-Hill Companies. |
Phosphorous Cycle
Phosphorous is an essential nutrient for plants and animals, but is not very common in the biosphere. This nutrient is found in soil and rock, but does not enter the atmosphere. Geologic processes bring phosphorous-laden ocean sediments to land, and weathering spreads them throughout the terrestrial environment. Plants absorb the phosphates from the soil and turn them into organic compounds which are consumed through the food chain. Decayed plant and animal matter return phosphates to the soil or to rock, and weathering also returns them to the ocean. The phosphorous cycle is one of the slowest of the biogeochemical cycles.
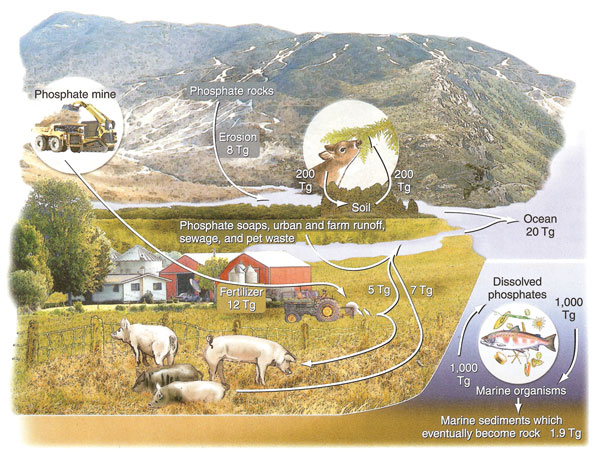 |
| Figure 2.27: Phosphorus cycle. Source: Author - Cunningham, W., et. al. 2007; Publisher - McGraw-Hill, N.Y. with permission of The McGraw-Hill Companies. |
Sulfur Cycle
Sulfur is an important component of proteins and enzymes in plants and the animals that feed on them. Most of the earth’s sulfur is found in ocean sediments. It is also found in the atmosphere as a result of evaporation, organism decay, volcanic eruptions, and as a product of industrial processes. Sulfur returns to the earth as particulate matter or acid deposition to be absorbed by plants and returned to the cycle.
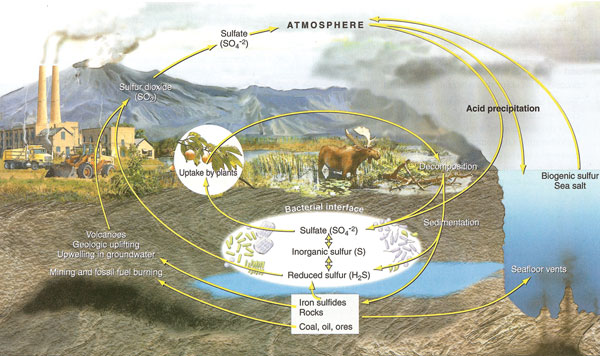 |
| Figure 2.28: Sulfur cycle. Source: Author - Cunningham, W., et. al. 2007; Publisher - McGraw-Hill, N.Y. with permission of The McGraw-Hill Companies. |
Hydrologic Cycle
The hydrologic cycle describes the movement of water above, on, and below the surface of the Earth. Water can be liquid, vapor, or ice at various places in the water cycle. Most of the exchange in the water cycle occurs through evaporation from oceans and precipitation back to oceans. About one-tenth of water evaporation from oceans falls over land, is recycled through terrestrial systems, and drains in rivers back to the oceans. Solar energy evaporates water and winds distribute water vapor across the earth. Water condenses and falls as precipitation (rain and snow) to support all terrestrial ecosystems. Living organisms return moisture—through respiration or perspiration—back into the atmosphere or into lakes and streams through which it eventually returns to the oceans. Water is responsible for the metabolic processes within cells, for maintaining the flow of key nutrients through ecosystems, and countless other important life sustaining services.
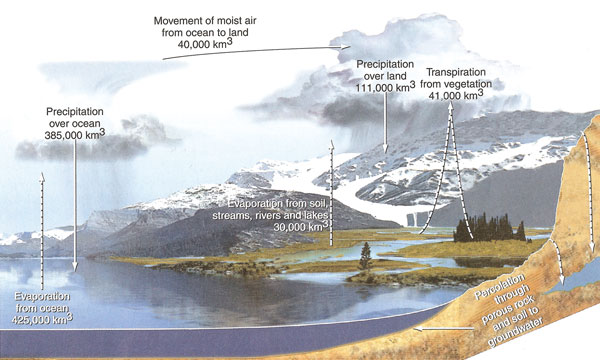 |
| Figure 2.29: Hydrologic cycle. Source: Author - Cunningham, W., et. al. 2007; Publisher - McGraw-Hill, N.Y. with permission of The McGraw-Hill Companies. |